This is a guest post by Harry Mercer. It tells about the best practices in Sulphur dyeing for fashion denim and was a part of the presentation made at Dhaka by him on a seminar on Sulphur Dyes by Fenazol
The Nature of Sulfur Dyes
- Sulfur dyes are a form of vat dyes – they are water-insoluble and in order to apply to fibers must be made water-soluble through the process of reduction.

- Reduction is a chemical process in which hydrogen is liberated. The hydrogen reacts with the dye and permits a water molecule to attach to the dye. The dye is transported into cotton fiber by the water.
- This reduced dye must then be oxidized. Oxygen reacts with the hydrogen producing water. Removing the hydrogen makes the dye insoluble, which results in the dye becoming physically trapped inside the fiber.
The Uses of Sulfur Dyes
- Sulfur dyes are commonly used for black, blue, brown, khaki and green colors.
- Sulfur dyes are applied to cellulosic fibers and blends of cellulosics with polyester, nylon and acrylics.
- Sulfur dyes can be applied with little difficulty and with excellent results at a relatively low cost.
- With good application methods, sulfur dyes are extremely wash-fast, however, they are usually not fast to chlorine bleaching.
Dark and medium colors are the most common with sulfur dyeing. Light colors can be produced by special procedures involving blended reducing agents, blends of surfactants, controlled oxidation and preparation methods like Mercerizing and bleaching.
Dark, dull sulfur dyeing
•Sulfur blacks are commonly applied in the 1st box on Indigo machines.
•The sulfur black dye molecule exists as very large polymers which require high temperatures for penetration into fibers (90-95 degrees).
•The maximum amount of sulfur black dye that can be efficiently applied is 4%on weight of cotton of a pure sulfur powder or 20% of a standard liquid (20% dye strenght). Darker sulfur blacks can be achieved by adding a sulfur blue.
•Sulfur blacks are best oxidized with an air passage like Indigo.
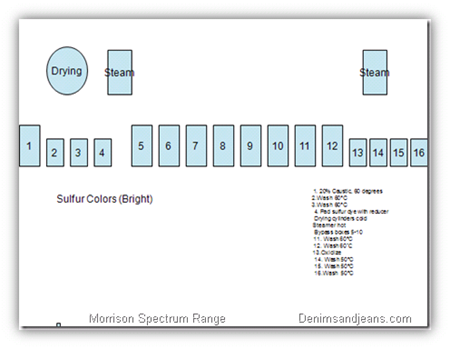
Boxes :
1. 20% Caustic, 60 degrees
2.Wash 60°C
3.Wash 60°C
4. Pad sulfur dye with reducer
Drying cylinders cold
Steamer hot
Bypass boxes 5-10
11. Wash 60°C
12. Wash 60ËšC
13.Oxidize
14. Wash 50°C
15. Wash 50°C
16.Wash 50°C
Light Sulphur Colors
- Sulfur dyeing procedures have been used to create a full range of colors including browns, yellows, greens, violets, reds, orange and neutrals (light/medium greys)
- Producing special colors with these processes involves the use of true sulfur dyes alone or in combination with sulfurized vats or special direct dyes that can be applied with special sulfur dyeing procedures.
- For success in fashion markets with these colors, marketing is focused on color “themes†such as “earth tonesâ€, “urbanâ€, sport or university colors.
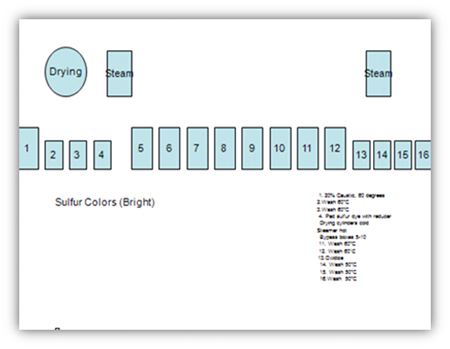
Control Of Light Sulphurs
- In order to match specific colors, dyes with greatly different affinity factors are mixed.
- Different affinity factors result in these dyes being picked up at different rates, so special methods are necessary to avoid color variation.
- Sulfur colors should be applied from 30 to 60 degrees to avoid color variation and must be chemically oxidized.
1. 20% Caustic, 60 degrees
2.Wash 60°C
3.Wash 60°C
4. Pad sulfur dye with reducer
Drying cylinders cold
Steamer hot
Bypass boxes 5-10
11. Wash 60°C
12. Wash 60ËšC
13.Oxidize
14. Wash 50°C
15. Wash 50°C
16.Wash 50°C
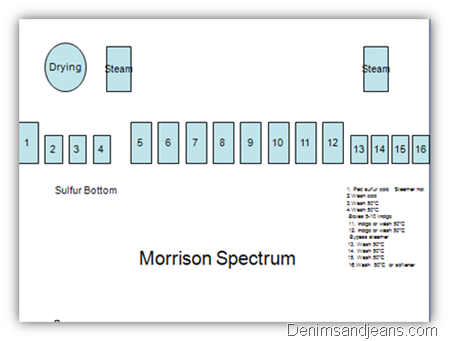
Sulphur Bottoming
- Bottoming of Indigo dyed yarns produces a darker, brighter effect with Indigo.
- The purpose of bottoming originally was to produce a darker color with less Indigo.
- While large amounts of sulfur dye can be applied in the 1st box, most of the sulfur is removed by hydrosulfite in the Indigo dye.
- Unlike other sulfur dye applications, a pH of 12 with sodium hydroxide is helpful to retard the dyeing for color consistency.
- A temperature of 60 degrees or less should be used to lower the affinity factor for consistent color.
Problems With Sulphur Bottoming 
- Sulfur bottoming can provide lighter shades because most of the dye applied in the 1st box is removed by the hydrosulfite in the Indigo boxes.
- The affinity factor (rate of dyeing) at high temperatures of sulfur dyes results in shade variation.
- If sulfur black is applied as a bottom, the Indigo boxes are contaminated with a yellow compound which causes discoloration of pure Indigo shades. Sulfur blacks are typically produced by polymerizing 2,4 –dinitrophenol(reddish-yellow) with sulfur. Hydrosulfite in the Indigo boxes-reduces the sulfur black to this yellowish compound.
- Sulfur bottoming should be conducted at temperatures of 30-60 degrees for consistency. Unlike other sulfur dyeing which should be conducted at pH 11, sulfur bottoming can be conducted better at pH 12 with sodium hydroxide which acts as a retarder to slow the rate of dyeing.
Boxes
1. Pre-wet 2% caustic 90°C
2.Wash 60°C
3.Wash 60°C
4. Wash cold
By pass drying cylinders
Bypass steamer
Boxes 5-10 Indigo
11. Wash 60°C
12. Pad sulfur topping
Steamer hot
13. Wash cold
14. Wash 50°C
15. Wash 50°C
16.Wash 50°C/Softener
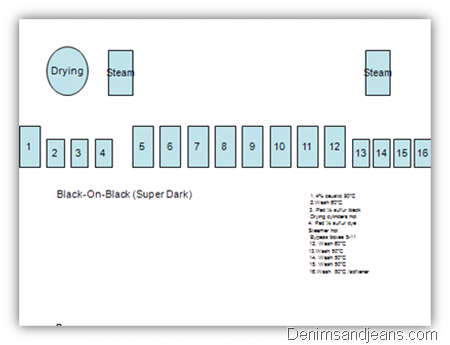
Black-on-Black Dyeing 
- Sulfur black dyes are low-intensity dyes that require large amounts for dark shades.
- The amount of sulfur black that will produce the darkest shade when applied properly is 4% of a 100% powder or 18% of a 20% liquid.
- It is difficult for cotton to easily absorb that quantity of dye in a way that produces a very dark shade.
- One method that has been used to produce darker, more colorfast sulfur blacks on yarn is to apply 50% of the dye, then dry the cotton, air oxidize and without washing, proceed to a second dye-box where the dye is applied again, dried oxidized with air again and washed.
Boxes:
1. 4% caustic 90°C
2.Wash 60°C
3. Pad ½ sulfur black
Drying cylinders hot
4. Pad ½ sulfur dye
Steamer hot
Bypass boxes 5-11
12. Wash 60°C
13.Wash 50°C
14. Wash 50°C
15. Wash 50°C
16.Wash 50°C /softener
Reducing Agents For Sulphur Dyes
- Because sulfur dyes cannot be dissolved in water directly, reducing agents must be utilized to produce hydrogen which allows the dye to attach to water.
- Any standard reducing agent can be used to dye sulfurs including reducing sugars (dextrins), sodium hydrosulfite, sodium bisulfite, sodium sulfide, sodium polysulfide and sodium hydrosulfide.
Selection of reducing agents
- Reducing agents for sulfur dyes should be selected according to the type of dyeing (batch or continuous), temperature of dyeing (cold or hot) and depth of color (light or dark).
- The reduction potential measured in millivolts for continuous sulfur dyeing as on Indigo machines should be at least -600 for dark, dull shades and at least -700 for bright, colorfast shades.
- The best sulfur dyeing for bright, consistent and colorfast shades requires blends of reducing agents.
Advances in sulfur dyeing
- Cold dyeing of sulfurs is possible on Indigo machinery in a full range of colors.
- This procedure has long been utilized by craft dyers in Asia.
- Cold dyeing of sulfurs have the advantage of requiring only 50% as much sulfur dye,
- Energy savings, excellent colorfastness, no polluting dye waste and less cotton damage for a soft touch and higher weaving efficiency.
Special Notes
- Dyeing with 100% sulfur blacks on yarn results in lower weaving efficiency. Normal yarn break levels with black can be achieved with improved washing and the use of buffers in dyeing.
- Sulfur blacks can be dyed with special procedures that provide wet rub-fastness of 4 and good washfastness even after 20 launderings.
Caution And Care
- The recommendations of many sulfur dye suppliers result in dye waste of 50% or more, which increases dyeing costs by twice that is necessary. Also, most of the additional chemicals used in sulfur dyeing, especially for blacks, are unnecessary.
- The use of fixatives after sulfur bottoming are often recommended to increase dye shades, but these are destroyed by the hydrosulfite in the Indigo boxes.
For more details on sulphur dyeing , check the site of FENAZOL
 This is a guest post by Harry Mercer. Mr. Mercer has 30 years experience in the denim business including 3 prominent U.S. denim companies. He is an expert colorist for measurement and color matching as well as textile testing.
This is a guest post by Harry Mercer. Mr. Mercer has 30 years experience in the denim business including 3 prominent U.S. denim companies. He is an expert colorist for measurement and color matching as well as textile testing.
Sponsored Link :