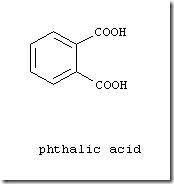
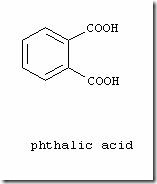
This is a technical writeup on various types of dyes used in Denim Dyeing by one of our guest writers – Adnan
Indigo Dyes
Background
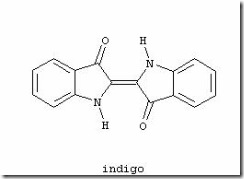
Indigo, or indigotin, is a dyestuff originally extracted from the varieties of the indigo and woad plants. Indigo was known throughout the ancient world for its ability to color fabrics a deep blue. Egyptian artifacts suggest that indigo was employed as early as 1600 B.C. and it has been found in Africa, India, Indonesia, and China.
The dye imparts a brilliant blue hue to fabric. In the dying process, cotton and linen threads are usually soaked and dried 15-20 times. By comparison, silk threads must be died over 40 times. After dyeing, the yarn may be sun dried to deepen the color. Indigo is unique in its ability to impart surface color while only partially penetrating fibers. When yarn dyed with indigo is untwisted, it can be seen that the inner layers remain uncolored. The dye also fades to give a characteristic worn look and for this reason it is commonly used to color denim. Originally extracted from plants, today indigo is synthetically produced on an industrial scale. It is most commonly sold as either a 100% powder or as a 20% solution.
History
The name indigo comes from the Roman term indicum, which means a product of India. This is somewhat of a misnomer since the plant is grown in many areas of the world, including Asia, Java, Japan, and Central America. Another ancient term for the dye is nil from which the Arabic term for blue, al-nil, is derived. The English word aniline comes from the same source.
The dye can be extracted from several plants, but historically the indigo plant was the most commonly used because it is was more widely available. It belongs to the legume family and over three hundred species have been identified. Indigo tinctoria and I. suifruticosa are the most common. In ancient times, indigo was a precious commodity because plant leaves contain only about small amount of the dye (about 2-4%). Therefore, a large number of plants are required to produce a significant quantity of dye. Indigo plantations were founded in many parts of the world to ensure a controlled supply.
Demand for indigo dramatically increased during the industrial revolution, in part due to the popularity of Levi Strauss’s blue denim jeans. The natural extraction process was expensive and could not produce the mass quantities required for the burgeoning garment industry. So chemists began searching for synthetic methods of producing the dye. In 1883 Adolf von Baeyer (of Baeyer aspirin fame) researched indigo’s chemical structure. He found that he could treat omega-bromoacetanilide with an alkali (a substance that is high in pH) to produce oxindole. Later, based on this observation, K. Heumann identified a synthesis pathway to produce indigo. Within 14 years their work resulted in the first commercial production of the synthetic dye. In 1905 Baeyer was awarded the Nobel Prize for his discovery.
At the end of the 1990s, the German based company BASF AG was the world’s leading producer, accounting for nearly 50% of all indigo dyestuffs sold. In recent years, the synthetic process used to produce indigo has come under scrutiny because of the harsh chemicals involved. New, more environmentally responsible methods are being sought by manufacturers.
Raw Materials
The raw materials used in the natural production of indigo are leaves from a variety of plant species including indigo, woad, and polygonum. Only the leaves are used since they contain the greatest concentration of dye molecules. In the synthetic process, a number of chemicals are employed as described below.
SOURCES AND USES: –
A variety of plants, including woad, have provided indigo throughout history, but most natural indigo is obtained from those in the genus Indigofera, which are native to the tropics. In temperate climates indigo can also be obtained from woad (Isatis tinctoria) and dyer’s knotweed (Polygonum tinctorum), although the Indigofera species yield more dye. The primary commercial indigo species in Asia was true indigo (Indigofera tinctoria, also known as Indigofera sumatrana). In Central and South America the two species Indigofera suffructicosa and Indigofera arrecta (Natal indigo) were the most important.
Natural indigo was the only source of the dye until about 1900. Within a short time, however, synthetic indigo had almost completely superseded natural indigo, and today nearly all indigo produced is synthetic.
In the United States, the primary use for indigo is as a dye for cotton work clothes and blue jeans. Over two billion pairs of jeans around the world are dyed blue with indigo. For many years indigo was used to produce deep navy blue colors on wool.
Indigo does not bond strongly to the fiber, and wear and repeated washing may slowly remove the dye.
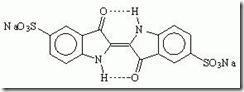
Indigo is also used as a food coloring, and is listed as FD&C Blue No. 2. The specification for FD&C Blue No. 2 includes three substances, of which the major one is the sodium salt of Indigotindisulfonate.
Indigotinesulfonate is also used as a dye in renal function testing, as a reagent for the detection of nitrates and chlorates and in the testing of milk.
CHEMICAL PROPERTIES: –
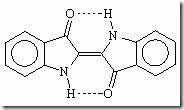
Indigo is a dark blue crystalline powder that melts at 390°–392°C. It is insoluble in water, alcohol, or ether but soluble in chloroform, nitrobenzene, or concentrated sulfuric acid. The chemical structure of indigo corresponds to the formula C16H10N2O2.
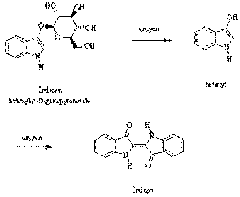
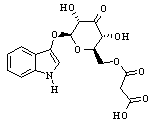
The naturally occurring substance is indican, which is colorless and soluble in water. Indican can easily be hydrolyzed to glucose and indoxyl. Mild oxidation, such as by exposure to air, converts indoxyl to indigo.
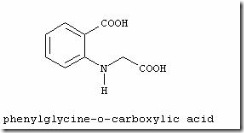
The manufacturing process developed in the late 1800s is still in use throughout the world. In this process, indoxyl is synthesized by the fusion of sodium phenylglycinate in a mixture of sodium hydroxide and sodamide.
Several simpler compounds can be produced by decomposing indigo; these compounds include aniline and picric acid. The only chemical reaction of practical importance is its reduction by urea to indigo white. The indigo white is reoxidized to indigo after it has been applied to the fabric.
Indigo treated with sulfuric acid produces a blue-green color. It became available in the mid-1700s. Sulfonated indigo is also referred to as Saxon blue or indigo carmine.
Tyrian purple was a valuable purple dye in antiquity. It was made from excretions of a common Mediterranean Sea snail. In 1909 its structure was shown to be 6,6′-dibromoindigo. It has never been produced synthetically on a commercial basis.
INDIGO CARMINE
HOW THE DYE WAS PRODUCED IN INDIA
The cut plant is tied into bundles, which are then packed into the fermenting vats and covered with clear fresh water. The vats, which are usually made of brick lined with cement, have an area of about 400 square feet and are 3 feet deep, are arranged in two rows, the tops of the bottom or "beating vats" being generally on a level with the bottoms of the fermenting vats. The indigo plant is allowed to steep till the rapid fermentation, which quickly sets in, has almost ceased, the time required being from 10-15 hours. The liquor, which varies from a pale straw colour to a golden-yellow, is then run into the beaters, where it is agitated either by men entering the vats and beating with oars, or by machinery. The colour of the liquid becomes green, then blue, and, finally, the indigo separates out as flakes, and is precipitated to the bottom of the vats. The indigo is allowed to thoroughly settle, when the supernatant liquid is drawn off. The pulpy mass of indigo is then boiled with water for some hours to remove impurities, filtered through thick woollen or coarse canvas bags, then pressed to remove as much of the moisture as possible, after which it is cut into cubes and finally air-dried Nature 1 November 1900
Indigo dye is an important dyestuff with a distinctive blue color (see indigo). The natural dye comes from several species of plant, but nearly all indigo produced today is synthetic. Among other uses, it is used in the production of denim cloth for blue jeans. The form of indigo used in food is called "indigotine", and is listed as FD&C Blue No. 2.
PHYTOCHEMISTRY OF INDIGO
Now just a bit of chemistry about how the compounds in plants are converted into indigo…
In Indigofera species, the precursor of indigo is indican
(left), indoxyl-β-D-glucopyranoside,
While in woad it is mainly isatan A (right)
INDUSTRIAL SYNTHESIS OF INDIGO
Current world production of indigo is 17,000 tons/year, mostly (40%) produced by BASF in Ludwigshafen. That is where things started in July 1897, using a process developed by von Heumann. It started with naphthalene…
VAT DYES
Vat dyes are used in the dyeing and printing of all types of cellulose fibres, and also of blends of cotton with polyester. In their coloured form they are insoluble pigments, so their applications depend on reversible reduction-oxidation (redox) reaction. In dye bath the pigment is converted into a water-soluble form using a strongly alkaline solution of a powerful reducing agent. This form the sodium “leuco†compound of the dye, which is soluble in water but often different in colour from the original in pigment. It is then allowed to dye the cellulose in this water-soluble from once exhaustion is completed the leuco compound is oxidized.
REDOX REACTIONS
Oxidation-reduction reactions (redox reaction) are important in textile colouration because they are an essential part of the process of the application of vat and sulphur dyes. In rather oversimplified terms, when a compound id oxidized it gain oxygen when something is reduced it loses oxygen. Reduction of a substance can also be thought of as gaining oxygen atoms, and oxidation as losing hydrogen atoms. For example when hydrogen reacts with oxygen to form water the hydrogen to become oxidized and the oxygen is reduced. In a redox reaction there is always a compound acting a reducing agent (hydrogen in this example). The reducing agents become oxidized the reaction by the compound that is being reduced, which is acting as an oxidizing agent. (In this case oxygen)
Sodium Sodium Hydroxide
Hydroxide ion ion
In the water molecule each hydrogen atom shares the only electron it possesses by pairing with one of the six electron of the oxygen atom, to form a covalent bond. Thus the hydrogen atom has lost one electron to become oxidized and the oxygen atom is reduced by gaining electrons. This is a more general way of expressing the phenomena of oxidation and reduction.
Oxidation = entails the loss of electrons by the oxidized compound
Reduction = entails the net gain of electrons by the reduced compound
vat dyes such as indigo and compounds derived from anthrequinone are applied after the temporary reduction of two carboxyl group in a conjugated chain, this convert the dye in to a colorless water insoluble form. The conversion is carried out using a strong reducing agent and in this reaction the two oxygen atom becomes reduced –O – and the two hydrogen atoms are oxidized to H+. The reduced form is called the leuco vat acid, and is applied from an alkaline. Once on the fibre it can be re -oxidized back to the insoluble carbonyl form by air or by the use of an oxidizing agent.
Sulphur dyes are also applied using a redox reaction mechanism, in which sodium sulphide is used as the reducing agent,
CHEMICAL NATURE OF VAT DYES
A large majority of vat dyes are based on the anthraquinonoid or the indigo (or thioindigo) chromophores; indigo, one of the oldest dyes still in use, remains popular through the wide use of indigo-dyed anthrequinone dyes are complex polycyclic quinines (Appendix 1), and they all possess two carbonyl groups (C = O) linked by alternate single and double bonds in a conjugated chain. The molecular arrangement is responsible for the easily reversible redox reactions on which the application of vat dyes depends.
In earlier countries, when all textile colorants where obtained from natural sources, indigo plant is steeped in a large vat. It is forming this ancient vatting process that the term fermentation vat dyes are derived. Fermentation converts one of the plant constituents into the soluble leuco dye, which diffuses out of the plant. The replacement of the natural by synthetic indigo at the end of the nineteenth century gave the imputes to research on other syntheses vat dyes have since followed.
Synthetic vat dyes are costly because they are difficult to prepare, so their use is usually directed to the higher quality fabrics. Nevertheless, they are widely used and noted for their high fastness to light, in the dyeing of fabric for uses such as awning, curtains, upholstery, military and naval uniforms and high quality gabardines.
High fastness to bleaching is another strong point of the anthraquinonoid group. This is exploited in the production of patterned fabric from vat-dyed yarn in which the white areas of the can be safely bleached out after weaving.
Anthraquinonoid vat dyes are widely used in the manufacturer of for example good quality, shirting’s, table cloths, towels, sportswear, high quality over all, fabrics of women and children clothing and tropical suiting, and yarns and effect threads where repeated washing will be required. With careful dye solution, the use of vat dyes allows is that the range lacks scarlet, maroon and wide shades.
APPLICATION OF VAT DYES
Both the extent of reduction and the rate at which equilibrium between the reduced and oxidized from is achieved are of practical significance. Vat dyes vary in the speed with which they go undergo reduction (Na2S2O4), which is capable of completely reducing even the most stable of vat dyes. As a result any difficulties in vatting can be over come by raising the vatting temperature, increasing the concentration of reducing agent or prolonging the vatting time. The vatted dyes must be kept in a strongly alkaline solution, because its leuco from is an insoluble acid. If instead of being formed as its water soluble salt, it will not readily oxidize to the coloured form.
There are still some application methods in which reduction and adsorption in the fibre take place rapidly and almost simultaneously. Under those conditions the rate and extent of reduction can be decisive factors in the dye stuff choice for example, in textile printing. Premature oxidation of the leuco compound in his print paste must also be avoided during both storage and steaming. This condition is usually met by using as the reducing agent sodium formaldehyde sulphoxylate (Formosul) a compound that is fairly stable in air at room temperature and develops the necessary action during steaming.
Variables such as pigment practical size and crystalline from can affect the rate of reduction but these are controlled by the dye manufacturing. Consequently the colourist needs to concentrate only on the temperature and the concentration of reducing agent.
Leuco compounds can be applied by batch wise methods similar to those used for other dye classes, but there are difficulties in obtaining leveling dyeing. The necessary high concentration of sodium hydroxide and reducing agent affect the exhaustion. The difference is that the option of reducing the concentration of the addition is not available, because they are needed to form the leuco compound. Serious leveling problems can be lessened by raising the temperature of dyeing and then cooling to obtain satisfactory exhaustion, or by using a dye bath auxiliary that restrains the rate of exhaustion. A different approach is to use specially formulated to stable dispersion of the pigment, in which form they are evenly distributed on the fabric by padding (pigment padding), followed by working the padded fabric through a of caustic soda and sodium dithionite on a jig. Pre-pigmentation can be carried out using package, jig, winch or beam dyeing machines provided specially formulated vat dyes are used. Once the pigmentation is completed, sodium hydroxide and sodium dithionite are added to the dye bath to reduce the pigment and allow the leuco day salt to diffuse into the fibre.
Oxidation in air or in solution is then used to regenerate the pigment. A soaping treatment is given to the fabric at the end of the dyeing process this is essential, both for cleaning the dyed fabric and for developing the final shade. In some cases a change in the crystalline form of dye accompanies the change in shade.
SOLUBILISED VAT DYES
The need to reduce vet dyes before use makes their application a cumbersome process. Although it is possible to isolate the reduced form of the dye. It is too readily oxidized in air for the manufacturer to provide the dyer with the leuco compounds. it is possible however to convert the leuco acid into the leuco ester, a derivative that has greater resistance to oxidation and grater solubility in water. Such ester can be formed by the reaction of a hydroxyl group of a leuco acid with sulphuric acid forming a sulphuric ester. The sodium salt of such esters are stable and can be stored until required for use. Since the ester group is only weakly attached to the rest of the dye molecule, it is easily removed by the action of sodium nitrate in dilute sulphuric acid. The regenerated leuco compound may then be oxidized back to the pigment form.
Solubilised vat dyes are less rapidly taken up than are the more conventional vat dyes and are mainly used for the production of pale shade. As with ordinary vat dyes application under alkaline condition is essential, thus eliminating wool from the list of possible substrates because alkaline condition modifies the wool fibres. The low up take and higher cost of solubilised vat dyes make them uneconomical for deep shade however and for theses normal vat dyes alternative have to be used.
A interesting property of solubilised vat yes is their sensitivity to light in the solubilised state. This is used to produce “photographic†print on fabric.
![]() R – OH + H2S2O4 R – OH + H2S2O4 + H2O
R – OH + H2S2O4 R – OH + H2S2O4 + H2O
Leuco vat sulphuric sulphuric ester
Dyes acid
SULPHUR DYES
Deposition of insoluble pigments inside fibres may achieve more cheaply using sulphur dyes. But with these the shade gamut is restricted to back, mauves, olives, Bordeaux and reddish-browns. One of the earliest and best known sulphur dyes is Cl sulphur black 1, which is popular black with good fastness properties still in use today.
Like vat dyes, sulphur dyes are reduced and applied as soluble leuco compounds that need to be kept under alkaline condition, but sulphur dyes need only sodium sulphide to act as both alkaline and reducing agent. A simplified the version of the reaction is represented in following equation. The structure of the chromophores of sulphur dyes is complex and unknown. Again like vat dyes, these dyes can not be applied to wool without damage to the fibres due to the action of sodium sulphide on the cystine cross links. After exhausting the dye bath for approximately 1 hour at 60-90Co the fabric is thoroughly rinsed and exposed to the atmosphere, where oxidation generates the mechanically entrapped insoluble pigment.
One disadvantages of certain sulphur dyes (although, strangely, this problems appears to arise with the black shade only) is that dyed material stored under condition of high humidity and temperature can lose its nature strength. This is because inadequate washing-off after dyeing can lead to the slow generation of sulphuric acid in the fibre, arising from the presence of sulphur.
Sulphur dyes are used mainly in the dyeing of cellulose fabrics and in blend of cellulose with polyester, nylon and acrylic fibres. Typical application is for heavy drill fabrics. Corduroys, overalls, denim, awning and canvas. Limited quantities are also consumed in the colouration of silk, paper and more widely, leather.
![]() Ar’ – S – S- Ar’ Reduction with Na2S Ar’ – S – S- Ar’
Ar’ – S – S- Ar’ Reduction with Na2S Ar’ – S – S- Ar’
About the author: Adnan is a textile engineer and working with a reputed denim garment company in Pakistan.He is looking after new denim developments.