This is a guest post by Harry Mercer. This is a very technical description – read on if you are technically oriented..
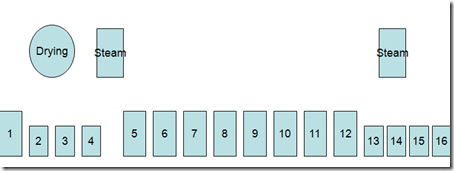
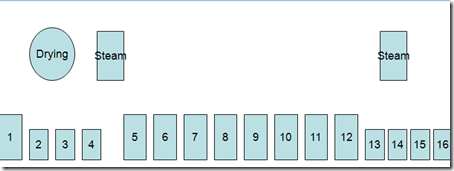
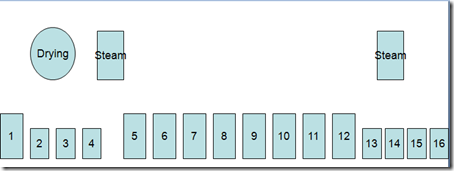
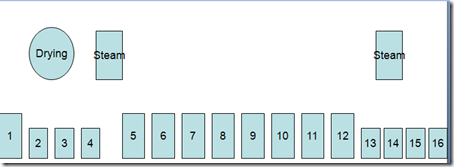
Here are given some important dyeing processes related to indigo dyeing – specially on Rope Dyeing .
Pre Treatment
Pre-treatment is conducted in the 1st tank. The most common pre-treatments are :
1. Sulfur bottoming
2. Scouring with sodium hydroxide
3. Causticizing or Mercerizing
Pre- Treatment : Sulphur Bottoming
•The purpose of sulfur bottoming was to:
- Originally to produce a dark shade on denim using less Indigo for lower costs
- In the U.S. sulfur bottoms were dyed using a combination of blue and black dyes
- In denim operations outside the U.S. the bottom is normally dyed with sulfur black
Pre – Treatment : Cotton Scouring
Cotton fibers contain impurities like waxes, pectins and minerals that will interfere with Indigo dyeing and result in streaks.-Sodium hydroxide at low concentrations (<5%) are applied at high temperatures (>85 C) in order to remove impurities and melt natural cotton waxes.
Pre – Treatment : Causticizing
- Causticizing generally refers to using sodium hydroxide at below Mercerizing concentrations (<18%).
- Cold causticizing of cotton yarn results infaster Indigo dye fading from laundryabrasion, darker Indigo color with the same % of Indigo and unique washdowns.
- Hot causticizing improves colorfastness
Pre – Treatment : Mercerizing
Mercerizing is the use of strong sodium hydroxide (18-30%) to swell surface fibers.
Caution:
- When using strong sodium hydroxide it is important to remove all of it.
- If sodium hydroxide is on the yarn as it enters the Indigo tanks, the yarn color will change.
- Concentrations of sodium hydroxide more than 18% are not a solution, but a gel and are difficult to remove.
- Mercerized yarns are more ring-dyed and dye more darkly than non-Mercerized.
- Mercerizing is normally conducted at low temperatures, but hot Mercerizing can be employed for a more abraded appearance after garment laundering.
Indigo Dyeing
- Indigo dyeing is unique and because of the complex chemical reactions should be correctly viewed a a form of chemical engineering.
- Only Indigo dyeing requires multiple dye applications for a dark shade.
- Color consistency of Indigo in recent decades has been unsatisfactory as a result of machine designs that do not apply basic principles of fluid mechanics properly and unstable dye mixes.
- Commonly, a single dye lot will have between 8 and 15 visually different shades from beginning to end and also have shade differences from one side to the other.
Indigo Dyeing Methods
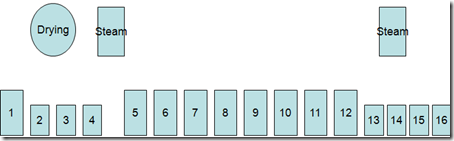
- Indigo dyeing follows the same basic steps regardless of machine design.
- Scour or dye bottoming in a heated tank,
- washing tanks, dyeing(1-20),a heated tank for topping (optional) and wash tanks.
- In different areas of the world,the same color is produced using 1.8, 2.0 or 4% Indigo depending on dyeing method.
|
Dark Indigo(1.8%) |
1. 15% caustic cold |
|
Stock Mix 80 g/l Indigo Pure 100 g/l 50% Caustic 70 g/l Hydro powder. |
Chemical Feed 120 g/l 50% caustic 60g/l Hydro powder Feed 1.4 liters per minute |
Dark Indigo Color
- This was an example of a typical method used in the U.S. for a very dark shade.
- In order to produce the same depth of color as 1.2% in the U.S., in Latin America 2.0% is used and in Asia from 2.4 to 2.8%.
- The U.S. method results in more surface (ring dyeing), which loses color faster.
Darkest Indigo Shades
- Very dark shades of Indigo are in demand currently around the world.
- Many companies use 4% or more Indigo on weight of yarn, which is expensive.
- 2% Indigo will produce the same depth if low levels of caustic are used(0-0.4%)
- For dark Indigo that does not lose color 2% applied normally, with an Indigo bottom.
Light Indigo Shades
- Dyeing Indigo in light shades results in a sky-blue impossible with any other dye.
- This is useful for shirting fabrics that are not strong enough for stonewashing, bleaching or cellulase treaments.
- Special procedures are necessary in order to avoid colorfastness problems.
|
Light Indigo 0.4% |
1. 4% caustic 90°C |
Control Of Sulphur Bottoming
- The typical methods used for dyeing sulfur bottoms result in denim shade differences.
- When applied as light colors, sulfur dyes should be dyed at temperatures <60 C,
- If dextrin reducing agents are used, which require 85 C, there will be variation.
- Sulfur bottoms are an exception to the normal pH for sulfurs(11), requiring 12.
|
Sulfur Bottom |
1. Pad sulfur(cold) |
Sulphur Topping
- In topping the sulfur dye is applied after the Indigo dyeing.
- Sulfur topping permits much darker color than a sulfur bottom, but is duller.
- Sulfur topping colors include black, blue-black, yellow brown and green.
- Sulfur toppings are used to produce slub appearances in normal yarn.
| Sulfur Top |
1. Pre-wet 2% caustic 90°C |
Reactive Dyes in Indigo Dyeing
- Reactive dyes can be applied on specially-Designed Indigo machines.
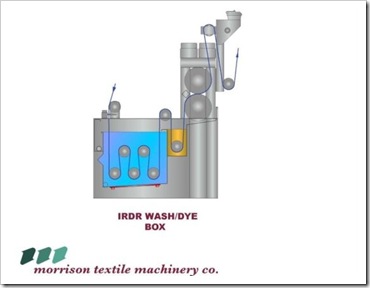
- Small 150 liter boxes are inserted inside the larger dye tanks for Indigo and sulfur.
- Steamers, drying units near the front of the machine and high-quality dye padders are required for quality dyeing.
|
Pad-Dry Chempad- Steam Reactives |
1. Pre-scour wetter plus chelate 90°C |
|
Pad Steam Reactive Topping |
1. Pre-wet 10% caustic 90°C |
Vat Dyeing
- Indigo and sulfurs are types of vat dyes.
- In non-denim cotton dyeing, another class of vats, anthaquinoids are used to produce a full range of colors that are colorfast.
- Some of these vat dyes can be blended with Indigo or applied using standard procedures on specially designed machines.
|
Pad-Dry Chempad Vats |
1. Pre-wet 4% caustic 90°C |
|
Pad-Dry Chempad Steam Vat Bottom |
1. Pre-wet 4% caustic 90°C |
Control Of Indigo Dyeing
The Indigo dyeing process begins with a concentrated mixture of Indigo, sodium hydroxide and reducing agent. This concentrated mixture (70-90 g/L Indigo) is delivered by pipes to the Indigo dye tanks where the dye concentration is reduced to 1-4 g/L for dyeing the cotton.
Dye Mixing Procedures
- Many denim companies find it difficult to control original and washed Indigo shades.
- The primary source of color differences is the instability and inconsistency of Indigo mixtures.
- As the concentration of reducing agent going to the dye machine changes, the color changes.
Uniform Indigo Mixtures
- For consistent Indigo dyeing, the mixture must have consistent concentrations of Indigo, sodium hydroxide and reducer from the top of the mixture to the bottom.
- The main cause of inconsistent Indigo mixtures relates to concentration levels.
- Instability of Indigo mixtures results from the decomposition of sodium hydrosulfite.
Consistency of Concentration
- There is a limit to the amount of any chemical that can be dissolved in water.
- When the limit of solubility of any chemical •In water is exceeded, precipitation occurs.
- Indigo mixes should not have more than 20% solids. At higher levels, chemicals and dye sink to the bottom of the tank.
Improving Dyeing Consistency
- When reducing agent sinks to the bottom of the tank, there is a higher concentration than in the top of the tank. As the dye enters the machine, the higher concentration results in a lighter, greenercolor and as the dye from the top of the tank enters the machine, the color is darker and redder.
Dye Control In Feeding Tank
- Stirring the tank for 2 minutes will improve dye uniformity between top and bottom.
- To avoid settling of dye and chemicals the total solids should not exceed 20%.
- The “glass plate†test can be used to test concentrations of hydrosulfite in the top and bottom. If dye requires 50 seconds to oxidize, there is about 50 g/L of reducer.
Buffers In Indigo Dyeing
- Alkaline buffers have been used to make very dark shades of Indigo with as little as 1% dye, more ring-dyed, faster fading.
- Reductive buffers can eliminate color differences in Indigo-dyed denims and can reduce hydrosulfite use by 30-50%.
Cold Dyeing Methods
- Sulfur colors can be dyed at low temperatures with specific buffers which produce more colorfast dyeings with no color variation.
- Cold dyeing methods have been used to blend Indigo and sulfurs, eliminating the need for separate bottoming and topping, while eliminating shade changes in both.
Special Dyeing Techniques
- On rope ranges, space dyeing techniques can be simple and produce a wide range of special effects in denim.
- By dyeing part of the yarns with a sulfur top and leaving the rest un-dyed, many companies produce a slub appearance with regular yarns.
- Blending ring yarns of different sizes also produces a slub appearance.
 This is a guest post by Harry Mercer. Mr. Mercer has 30 years experience in the denim business including 3 prominent U.S. denim companies. He is an expert colorist for measurement and color matching as well as textile testing.
This is a guest post by Harry Mercer. Mr. Mercer has 30 years experience in the denim business including 3 prominent U.S. denim companies. He is an expert colorist for measurement and color matching as well as textile testing.